Driving the adoption of hydrogen energy through advancements in functional materials and process engineering.
Hydrogen can be produced from a variety of primary energy sources, including renewables, and allows for the highly efficient extraction of electrical and thermal energy through devices such as fuel cells. As a secondary energy source, it enables large-scale transportation and long-term storage. However, because hydrogen exists as a gas at room temperature and standard pressure, it requires a massive volume to store. To overcome this challenge, hydrogen can be stored compactly by converting it into liquid chemical carriers or by absorbing it into solid-state materials as hydrogen atoms.
Based on the principles of material science, we are developing these hydrogen storage materials through precise nanoscale control.
Furthermore, because hydrogen energy utilization systems involve diverse combinations of production and extraction methods, the required storgae functions and processes vary significantly. Our goal is to propose highly-efficient hydrogen storage and supply processes tailored to specific applications, alongside the development of the advanced storage materials and containers necessary to realize them.
Magnesium (Mg)
Among hydrogen storage materials, Mg is an abundant and inexpensive resource that forms Mg hydride (MgH2) to store H2 at a high capacity (7.6 mass%, 109 kgH2 m−3). Although its inactive surface results in slow H2 absorption and desorption reaction rates, these rates have been improved by distributing an appropriate catalyst at the nanometer level [14]. Furthermore, to accommodate the volume changes and pulverization of MgH2 during H2 absorption and desorption, an MgH2-CNT composite has been fabricated. This structure supports catalyst-doped MgH2 particles within a three-dimensional sponge-like carbon nanotube (CNT) membrane, which provides both heat resistance and flexibility [49]. Moreover, focusing on the property that H2 possesses the highest thermal conductivity among gases, a novel method for H2 absorption and desorption has been proposed. This method is that these MgH2-CNT composites are set in parallel and low- or high-temperature H2 is circulated within the tank, and it is currently being evaluated from both experimental and numerical simulation [44, 55].
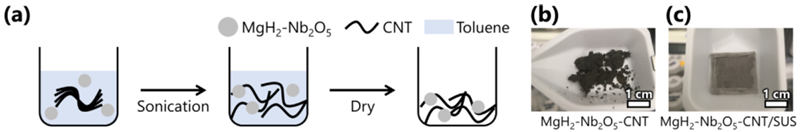
Fabrication process of the MgH2-Nb2O5-CNT composite[49]
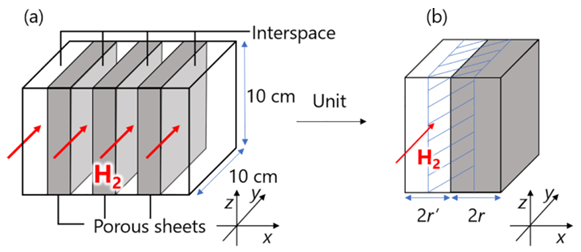
MgH2 sheet tank utilizing hydrogen as a heat transfer medium[44]
Current Main Research Themes
・Development of an MgH2 tank utilizing hydrogen as a heat transfer medium and design of an intergrated system with fuel cells/ water electrolysis
・Establishment of an in-air fabrication process for MgH2-carbon nanotube composite membranes with cycle durability
・Flow synthesis of Mg fine particles by arc discharge and evaluation of H2 storage properties
Metal Hydride
The hydrogen storage alloy LaNi5 is currently being developed for application in stationary hydrogen storage tanks. The alloy is initially in powder form, and it undergoes pulverization due to volume expansion and contraction of approximately 25% during hydrogen absorption and desorption. When this alloy is loaded into a tank, the pulverization effects the packing state inside the tank, resulting in an uneven supply of hydrogen and heat. This leads to a deterioration in the hydrogen absorption and desorption characteristics.
To address this issue, we have fabricated LaNi5-resin composite pellets by binding LaNi5 particles with a resin, successfully achieving both high hydrogen absorption and desorption performance as well as the preservation of the pellet shape. We evaluate the thermal conductivity characteristics of these pellets. Currently, we are advancing the design of alloy structures and alloy tanks that possess high thermal conductivity while maintaining a high volumetric hydrogen density.

LaNi5-resin pellets
Current Main Research Themes
・Development of a hydrogen storage alloy-polymer-expanded graphite structure for rapid hydrogen absorption and desorption
・Investigation of capacity enhancement by compositing metal hydride with metal-organic framework
Liquid ammonia(NH3)
Ammonia is an energy carrier with a gravimetric hydrogen density of 17.3 mass%. It is liquefied at approximately 1.0 MPa at room temperature, possessing a high volumetric hydrogen density of 108 kgH2 m−3. We are focusing on hydrogen generation from ammonia via electrolysis. Currently, our research primarily advances the electrolysis of ammonia in aqueous solutions[43]and the electrolysis of liquid ammonia[25, 51].
In both systems, the anodic reaction for nitrogen evolution exhibits a high overpotential, which has driven our efforts to develop novel electrocatalysts. Furthermore, to increase the surface area of these electrocatalysts, we are developing Metal-CNT membrane electrodes, where metal nanoparticles are supported on a three-dimensional sponge-like carbon nanotube (CNT) membrane. We are also conducting the improvement of Faradaic efficiency to enhance current density at low overpotentials, as well as the design of zero-gap cells.

Pt-CNT electrode[43]
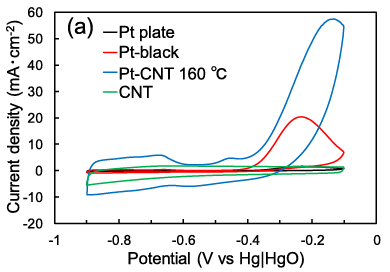
Anodic electrolysis characteristics of aqueous NH3 solutions
(Pt-CNT electrode)[43]
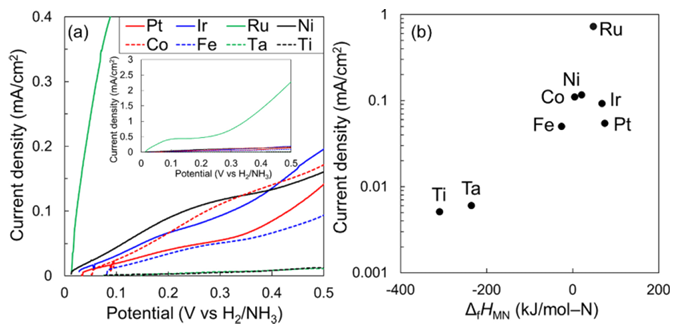
Anodic electrolysis characteristics of liquid NH3 (various metal plates)[51]
Current Main Research Themes
・Cathodic reaction mechanism and improvement of Faradaic efficiency and current density in liquid NH3 electrolysis
・Liquid NH3 electrolysis for hydrogen production using Ru-supported CNT membrane electrodes
・Electrolysis of NH3 in aqueous systems using Ir-supported CNT membrane electrodes
・Development of an integrated cell for NH3 electrolysis in aqueous systems
Other themes
We are also conducting further research related to energy materials and energy processes.
Current Main Research Themes
・Synthesis of nitrogen-containing organic compounds via electrolysis using aqueous carbonate solutions for CO2 fixation
・Hydrogen generation by electrolysis of low vapor pressure liquid ammonia adding inorganic solts
・Evaluation of CO2 absorption performance of MgO doped with eutectic ternary carbonate as a CO2 absorbent

